Article Text
Abstract
Background Pain at intrauterine contraception (IUC) fitting may be a deterrent for women and any reduction in discomfort would be likely to be beneficial to both women and clinicians.
Aim This study investigated the associated pain and the ease of use of two different stabilising forceps at the time of IUC placement.
Design Single-blinded randomised controlled trial. One hundred parous women aged 18–50 years who had undergone at least one vaginal delivery were recruited.
Setting An integrated sexual health service in the North of England.
Methods Women were randomised to the use of either a Littlewoods forceps or a single-toothed tenaculum forceps to stabilise the cervix at the time of IUC placement. Pain levels were measured at forceps application, IUC insertion, and 5 and 10 minutes post-procedure using a visual analogue scale (VAS). A clinician graded ease of use of the forceps and any associated bleeding. Wilcoxon rank-sum tests were used to compare pain levels between the two forceps groups. Linear regression was used when adjusting for potential confounders.
Results Mean VAS scores were similar at forceps application (p=0.52), IUC fitting (p=0.10) and at 5 minutes (p=0.32). There was a statistical difference in the degree of pain experienced at 10 minutes in the tenaculum group (p=0.01). Physicians found both forceps easy to use and there was no difference in bleeding (p=0.49).
Conclusions Parous women who have had a vaginal delivery can be reassured that IUC fitting is well tolerated. Pain scores were not affected by the type of forceps used to stabilise the cervix at IUC placement. Both forceps investigated were easy to use.
- intrauterine devices
- intrauterine systems
- statistics
Statistics from Altmetric.com
Key message points
There is no difference in pain or bleeding when either Littlewoods forceps or single-toothed tenaculum forceps are used to stabilise the cervix at intrauterine contraception (IUC) fitting.
Clinicians can use their forceps of choice when fitting an IUC in a parous woman who has experienced a vaginal delivery as the level of reported pain is low.
Introduction
There have been a number of published studies examining pain associated with insertion of intrauterine contraception (IUC).1–3 Anticipated pain is one of the main deterrents to women considering a copper intrauterine device (IUD) or hormonal intrauterine system (IUS).1 Brockmeyer et al.4 found that most women experience pain which is similar to menstruation in intensity, but a small number of women report more significant pain, particularly if they are nulliparous. In their study of nulliparous women, Marions et al.5 found that 17% reported severe pain at the time of IUC fitting compared with 11% of parous women in another study with similar methodology.6
Guidance from the Faculty of Sexual & Reproductive Healthcare recommends using forceps to stabilise the cervix when fitting IUCs in order to reduce the risk of uterine perforation.7 Some experts in the field have suggested that relatively atraumatic forceps such as the Allis or Littlewoods forceps may cause less pain and bleeding than forceps that pierce the cervical epithelium such as a single-toothed tenaculum.8 Most studies examining pain at IUC placement do not specify the type of forceps used. One published study investigated pain associated with different forceps but the numbers in the study were small and nulliparous as well as multiparous women were recruited.9
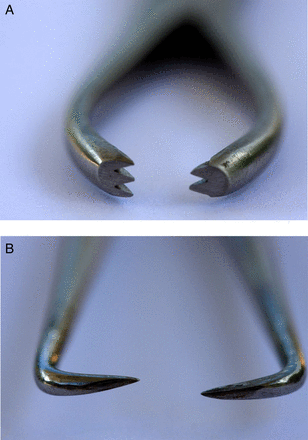
This study was designed to compare the ease of use and discomfort felt when two different types of forceps were used to stabilise the cervix at IUC fitting in women who had experienced a vaginal delivery. Figure 1A, B depicts the two forceps used in the study.
(A) View of tip of Littlewoods forceps. (B) View of tip of single-toothed tenaculum forceps.
Methods
We conducted a randomised, single-blind clinical trial investigating the pain associated with the use of either a single-toothed tenaculum or a Littlewoods forceps to stabilise the cervix for IUC fitting. The trial was conducted at an integrated sexual health clinic in the North of England between November 2013 and November 2014. Women aged 18–50 years, who had had at least one vaginal delivery, a negative Chlamydia trachomatis screen (if aged under 35 years) and who had regular menstrual cycles (between 21 and 35 days) were recruited.
Exclusion criteria included women who were nulliparous or who were parous but with no previous vaginal delivery, known or suspected pregnancy, a history of or active genital malignancy, acute cervicitis or vaginitis or current genital infection, post-abortion sepsis in the previous 3 months, a uterine cavity measurement of <6 cm or >10 cm, known structural abnormalities of the cervix or uterine cavity, submucous myomas or endometrial polyps, known endometrial hyperplasia, known hypersensitivity to latex or to components of the IUC, established immunodeficiency, recent trophoblastic disease with human chorionic gonadotrophin levels remaining elevated, history of chronic alcoholism, drug dependence or abuse, or severe psychological disorder.
Women were fitted with their chosen IUC, which was either a Mirena® IUS, Nova T380® IUD or a TT380® Slimline IUD.
The primary outcome was any difference in pain between use of either forceps at the time of their application. Secondary outcomes included:
Difference in pain experienced between the two types of forceps when the IUC was inserted and pain at 5 and 10 minutes post-procedure.
Difference in pain experienced between the two types of forceps after adjustment for:
taking oral analgesia before the procedure (pain at all four time points)
previous colposcopic treatment (pain at all four time points)
IUC removal prior to fitting the new device (pain at all four time points)
having had a more difficult IUC fitting (pain at time of IUC insertion, 5 and 10 minutes)
having had a prolonged IUC fitting (defined to be in the top 10% of times: pain at times 5 and 10 minutes).
Difference in pain experienced between:
women who did and did not take oral analgesia prior to the treatment (pain at all four time points)
women who had and had not previously had a colposcopic treatment (pain at all four time points)
women who did and did not have an IUC removed prior to fitting the new device (pain at all four time points).
Difference in:
level of bleeding associated with the use of the different forceps
the number of times the forceps was reapplied before the cervix was stabilised
ease of application of the forceps.
Potential participants who met the eligibility criteria were recruited on the day of their procedure. Participant recruitment, enrolment and IUC fitting were carried out by two of the authors (JMMS, KMC). All participants were informed of the purpose of the study and gave their written consent. A pre-procedure questionnaire recorded age, parity, ages of living children, mode of past deliveries, lactation status, current contraception, last menstrual period, whether any analgesia had been taken on the day of IUC fitting, and history of any past cervical treatments (e.g. large loop excision of the transformation zone of the cervix).
Participants were equally allocated to one of two groups using a computer-generated fixed-block randomisation scheme created by the statistician. Randomisation allocations were concealed in sealed envelopes numbered from 1 to 100 and opened by the physician just before the fitting procedure.
Before the procedure participants were shown how to use a sliding wooden 100 mm visual analogue scale (VAS) (0 cm=no pain, 10 cm=worst pain imaginable). VAS has been shown to be a reliable way to assess the experience of acute pain, and has been used in previous trials examining pain associated with IUC insertion.10
Immediately following forceps application to the anterior lip of the cervix the VAS pain scale was recorded by the patient. VAS scores were then collected immediately after the IUC was inserted, and at 5 and at 10 minutes post-insertion. All VAS scores were documented by the healthcare assistant (HCA) who was present during the IUC fitting. The HCA was also responsible for timing the procedure. None of the participants was given any local anaesthetic during the IUC fitting procedure.
The physician scored the ease of application of the forceps using the following categories: easy; difficult; very difficult – would have preferred to use another instrument. The physician also scored whether the forceps required re-application in the following categories: no; yes – once; yes – more than once. The physician categorised the ease of removal of the forceps as either easy or difficult. The amount of bleeding from the forceps application site was scored using the sliding VAS from no bleeding (0 cm) to excessive bleeding (10 cm). The ease of insertion of the IUC was scored using the scale from easy (0 cm) to extremely difficult (10 cm).
The aim was to recruit 100 women into the study with no patient entering more than once. All 100 women who were recruited underwent a successful IUC fitting. A power calculation was not performed as no clinical or statistical information was available at the time of designing the study to differentiate between the two chosen forceps.
Research ethics committee approval was obtained for the study from the Newcastle and North Tyneside Research Ethics Committee (Trial Reference Number 09/H0907/78).
The data were collated using a Microsoft Excel database. Continuous demographic variables were compared between the two groups using two-sided two-sample t-tests. Binary demographic variables were compared using a Chi-squared (χ2) test. Unadjusted comparisons between pain levels in the two forceps groups at each of the four time points were carried out using a two-sample Wilcoxon rank-sum test. The continuous outcomes were analysed using linear regression, and logistic regression was used for binary outcomes. For the secondary outcomes, p values were adjusted using a Bonferroni correction to adjust for multiple comparisons. This corresponded to testing secondary outcome 1 against p=0.017 (0.05/3), secondary outcome 2 against p=0.003 (0.05/17), secondary outcome 3 against p=0.004 (0.05/12) and secondary outcome 4 against p=0.017 (0.05/3). Statistical analyses were carried out using the statistics software Stata V.13 (StataCorp LP, College Station, TX, USA).
Results
A total of 100 women were enrolled into the study and all were randomised to having either a single-toothed tenaculum or Littlewoods forceps applied to the cervix prior to IUC fitting. Analysis of socio-demographic characteristics showed no differences between the two groups (Table 1).
Baseline characteristics of study participants
Mean VAS pain scores were similar between the two groups at forceps application, at IUC insertion and at 5 minutes post-IUC insertion (Table 2). There was a statistically significant difference in pain scores at 10 minutes post-IUC insertion, but this difference is unlikely to be significant clinically as pain scores recorded at 10 minutes were so low. This difference was unaffected by the type of IUC fitted.
Mean visual analogue scale pain scores
After statistically adjusting separately for each of pre-medication with oral analgesia, history of colposcopic treatment to the cervix and undergoing an IUC removal before an IUC was fitted, there was no significant difference in pain scores between the tenaculum and Littlewoods forceps groups (Table 3).
Comparisons of pain scores between forceps after removing potential confounding factors
The cut-off for considering a procedure time to be prolonged was ≥122 seconds, which was the top 10% of times as defined in the Methods section. There was no significant difference in pain between the forceps used after adjustment for a prolonged procedure time at either 5 (p=0.510) or 10 minutes (p=0.072). Similarly, there was no significant difference in pain between the forceps used after adjustment for the physician finding the IUC insertion to be complex (at forceps application p=0.441; at insertion p=0.148; 5 minutes p=0.522; 10 minutes p=0.085).
There was no difference in pain between women who did and did not take oral analgesia pre-procedure, and between women who did and did not have an IUC removed prior to the new IUC being fitted. More pain was reported at forceps application by women who had previously undergone colposcopic treatment. This was not dependent on the forceps used and was significant even after the Bonferroni adjustment, with p=0.0037 (Table 4).
Effect of analgesia, previous colposcopic treatment and intrauterine contraception refit on visual analogue scale pain scores
Before adjusting for multiple testing it was statistically significantly easier to apply a single-toothed tenaculum than a Littlewoods forceps (p=0.040; odds ratio=0.19; 95% confidence interval 0.04–0.93). However, after applying a Bonferroni correction for multiple testing, this was not statistically significant (Bonferroni cut off for significance p=0.017). There was no difference between the forceps with regard to need for reapplication. There was no statistically significant difference in bleeding between the two groups (p=0.487).
Discussion
This study shows that pain scores are low and not statistically different when either a relatively atraumatic Littlewoods forceps or a single-toothed tenaculum is applied to stabilise the cervix at IUC fitting in women who have previously experienced a vaginal delivery. Pain scores were also low immediately following the IUC fitting and at 5 and 10 minutes post-insertion. Similar findings were found by Doty and MacIsaac.9 However, a strength of this study is that we investigated a more homogeneous group of women by excluding those who had never undergone a vaginal delivery, as pain associated with IUC placement is greater in that group.4 ,5 Our findings suggest that we can reassure women who have experienced a vaginal delivery that minimal pain is felt during IUC placement.
Additional strengths of this study are that it is a randomised trial and the participants were successfully blinded to the intervention. Only two experienced IUC fitters were involved in this study, thereby reducing inter-fitter variables.
Bahamondes et al.1 suggested that having a HCA present during an IUC fitting can decrease anxiety and in turn reduce a woman's experience of pain. Initially an attempt was made to use the same HCA for all procedures but this proved difficult to maintain throughout the trial. Five HCAs in total assisted with the IUC fitting, but an HCA was present at every fitting and this may have contributed to the low pain scores recorded.
A variety of oral analgesics was taken prior to the IUC fitting, but there is no reason to believe that the type of analgesia would have differed greatly between the two groups. This study confirms previous findings that those who took oral analgesia before the procedure did not have a statistically significant difference in pain scores from those who did not, and we therefore do not need to routinely recommend that women who have had a vaginal delivery should take oral analgesia before attending for an IUC fitting.11 ,12
Although there was no statistical difference after adjusting for multiple testing, we found that a single-toothed tenaculum grasped the cervical lip more effectively when the cervix was flat or flush with the vaginal vault, thereby providing better stabilisation of the cervix. Healthcare professionals can choose either forceps based on the clinical circumstances.
We found higher pain levels when either forceps was applied to the cervix of women who had undergone colposcopic loop diathermy treatment of the cervix. This warrants further investigation as there are no studies published in this area.
We found that there was no difference in bleeding between the two types of forceps. Doty and MacIsaac9 found that the single-toothed tenaculum caused more bleeding, resulting in a significantly longer procedure time for this group. As we did not find this effect in our study, we suggest that future studies might look at the differences in bleeding between different forceps in more detail.
A limitation to our study was the sample size of only 100 participants. Although more patients were recruited than in the Doty and MacIsaac9 study, we could not perform a power calculation to determine the sample size prior to starting our study due to the absence of published data. It should therefore be noted that 50 patients in each group may not be sufficient to draw firm conclusions as this number might not be large enough to show any definite effect. However, in view of the similarity of the results of the two groups in this study, it would be necessary for a very much larger study to be performed in order to demonstrate clinically significant differences in women's experience of pain.
Conclusion
IUC is a highly acceptable and effective contraceptive method with low discontinuation rates. This study shows that women who have experienced a vaginal delivery experience little pain when IUC is fitted and this cannot be further reduced by using a particular type of stabilising forceps. Both the relatively atraumatic Littlewoods forceps and single-toothed tenaculum forceps are easy for clinicians to use and appear to be associated with no difference in risk of cervical bleeding.
Footnotes
Competing interests Diana Mansour has received honoraria and travel expenses for lecturing and taking part in conferences and advisory boards from Aspen, Astellas, Bayer Healthcare, Consilient Health, HRA Pharma, MSD and Pfizer.
Patient consent Obtained.
Ethics approval Newcastle and North Tyneside Research Ethics Committee.
Provenance and peer review Not commissioned; externally peer reviewed.
Linked Articles
- Editorial
- Highlights from this issue
- Letters to the editor