Article Text
Abstract
Introduction Reduced funding to contraceptive services in the UK is resulting in restricted access for women. Pharmacists are already embedded in sexual and reproductive health (SRH) care in the UK and could provide an alternative way for women to access contraception. The aim of this study was to determine the views of UK contraception providers about community pharmacist-led contraception provision.
Methods An anonymous questionnaire was distributed to healthcare professionals at two UK SRH events, asking respondents about: (1) the use of patient group directions (PGDs) for pharmacist provision of oral contraception (OC); (2) the sale of OC as a pharmacy medicine or general sales list medicine; (3) the perceived impact of pharmacy provision of OC on broader SRH outcomes; and (4) if other contraceptive methods should be provided in pharmacies.
Results Of 240 questionnaires distributed, 174 (72.5%) were returned. Respondents largely supported pharmacy-led provision of all non-uterine methods of contraception, excluding the contraceptive implant. Provision of the progestogen-only pill by PGD was most strongly supported (78% supported initiation). Respondents felt that the use of bridging (temporary) contraception would improve (103/144, 71.5%), use of effective contraception would increase (81/141, 57.4%), and unintended pregnancies would decline (71/130, 54.6%); but that use of long-acting reversible contraception would decrease (86/143, 60.1%). Perceived barriers included pharmacists’ capacity and competency to provide a full contraception consultation, safeguarding concerns, and women having to pay for contraception.
Conclusions UK SRH providers were largely supportive of community pharmacy-led provision of contraception, with training and referral pathways being required to support contraception delivery by pharmacists.
- access to contraception
- contraception service provision
- pharmacist
- community pharmacy
- hormonal contraception
Statistics from Altmetric.com
- access to contraception
- contraception service provision
- pharmacist
- community pharmacy
- hormonal contraception
Key messages
The majority of UK contraception providers support community pharmacy-led provision of non-uterine contraception, excluding the contraceptive implant.
They strongly support pharmacists providing progestogen-only pills by patient group direction as part of their SRH service provision.
They oppose the reclassification of contraception to allow over-the-counter purchase as a general sales list medicine.
Introduction
In the UK, contraception has been available free of charge on the National Health Service (NHS) to all women for almost 50 years. The most recent UK National Survey of Sexual Attitudes and Lifestyles reported that >90% of women had obtained contraception within the last year, more than half of whom accessed this via their general practitioner (GP).1 Overall use of contraception in the UK has increased over the last decade, as has use of community clinics and retail outlets such as pharmacies for accessing contraceptive supplies.1 In addition, recent years have seen the advent of women being able to obtain or buy contraception on the internet, from online pharmacies and online sexual health providers.
With an increased variety of providers, it would seem logical that access might be improved as a result. However, there is a current dichotomy – aspirations to improve access to contraception2–4 are fighting against funding cuts to sexual and reproductive health (SRH) services.5 6 In 2017, the Advisory Group on Contraception reported that in the last 2 years, contraceptive services in over one-third of English local councils had been closed or were under threat.7 The services where women have traditionally accessed contraception (GP and SRH) are facing cuts, resulting in reduced appointment numbers, longer waiting times and restricted opening hours. Consequently, providers are concerned that this combination will lead to reduced access to contraception.
Pharmacy contraception provision has been highlighted as an area for development. Anonymity, long opening hours, no requirement for appointments, and a large number of services covering wide geographical areas allow community pharmacies to provide services at times and locations convenient to women. In the UK, contraception is a prescription-only medicine (POM) and can be supplied under instruction from a prescription or a patient group direction (PGD). Contraception is not currently available as a pharmacy medicine or general sales list (GSL) medicine.
Patient group direction (PGD): PGD is a legal mechanism enabling supply of a POM. It allows specified healthcare professionals (HCPs) to supply and/or administer a medicine directly to patients with identified clinical conditions without needing a prescription or instruction from a prescriber.8
Pharmacy medicines (P-medicines): People can buy medicines classified as P-medicines, but only under a pharmacist’s supervision. These medicines are not usually displayed on open shelves.9
General sales list (GSL) medicines: People can buy GSL packs from retail outlets (eg, supermarkets) and by self-selection in pharmacies.9
Community pharmacists are already embedded in SRH care in the UK, having provided emergency contraception (EC) for almost 20 years10 and chlamydia screening for over a decade.11 A small number of pharmacies also provide oral contraception (OC) as a bridging method after EC, or for initiation/continuation without EC.12–14
Previous studies have shown pharmacists are willing and clinically competent to provide OC12–15 and this is acceptable to women.12–14 16 17 A study of nearly 500 contraception providers in the United States showed almost three-quarters supported pharmacists initiating combined hormonal contraception (CHC), progestogen-only pills (POPs) and injectable contraception (IC),18 and a UK study of SRH clinicians showed that >90% felt positively about pharmacists initiating POP at the time of EC.19
The aim of this study was to determine the views of UK contraception providers about pharmacist-led provision of contraception.
Methods
The survey was conducted using paper-based, self-completed questionnaires, distributed at two UK SRH events in May 2018: the Faculty of Sexual & Reproductive Healthcare Annual Scientific Meeting (ASM, Birmingham) and NHS Lothian Sexual Health Update (LSHU, Edinburgh). These events provided the opportunity to reach a convenient sample of sexual health practitioners in a range of professional roles.
Permission was sought from the event organisers to distribute the survey, and ethical approval was not required as it was a health service evaluation. Information about the background and aims of the study were provided for participants, and consent assumed by means of questionnaire completion. Study participation was voluntary and anonymous.
The questionnaire was designed to elicit respondents’ views on four topics:
Pharmacists using PGD for POP and combined oral contraception (COC) (strongly support, support, neutral, oppose, strongly oppose)
Sale of POP and COC as a P-medicine or GSL medicine (strongly support, support, neutral, oppose, strongly oppose)
Impact of provision of POP and COC in community pharmacies on six broader SRH outcomes (improve, same, worse, unsure)
Whether other hormonal (non-uterine, non-oral) contraceptive methods should be provided in community pharmacies (yes, no, unsure)
The definition of PGD and the legal classifications of medicines by the Medicines and Healthcare products Regulatory Agency (MHRA) was provided.
A validated tool was not available, therefore the researchers developed the questions pragmatically to meet the study aims, considering previously reported concerns, barriers and opportunities of expanding contraception provision in pharmacies.18 The questionnaire included questions with fixed and rating scale responses, with opportunities for additional comments to be made as free-text responses. It was piloted with five HCPs and found to be comprehensive, with ample opportunity to provide additional comments.
Data collected from both events were collated into a Microsoft Excel database and analysed together. Statistical analysis was performed on the quantitative data and presented as descriptive statistics. The respondents’ comments were analysed and presented thematically to highlight key concerns, barriers and opportunities in relation to expansion of contraception provision in community pharmacies.
Patient involvement
As this study was investigating views of healthcare providers, patient involvement was not sought. A supplementary study is being undertaken to assess patients' views on pharmacy provision of contraception.
Results
A total of 240 questionnaires were distributed, with 174 questionnaires (102 from ASM, 72 from LSHU) returned, giving a response rate of 72.5%. table 1 shows the respondents’ demographics.
Characteristics of respondents
Pharmacy provision of POP
Respondents were asked if their local pharmacists were currently providing POP by PGD. Of 173 responses, 78 (45.1%) indicated ‘no’, 26 (15.0%) ‘yes’ and 69 (39.9%) ‘unsure’.
Respondents were asked if they supported or opposed PGD provision of POP by pharmacists (initiation and continuation) and the reclassification of POP as a P-medicine or GSL medicine. The results are shown in table 2. The vast majority of respondents supported PGD provision of POP for initiation and continuation (78.0% and 90.8%, respectively). The majority (59.0%) supported the reclassification of POP to a P-medicine; conversely, the majority opposed the reclassification of POP as a GSL medicine (63.3%).
Respondents’ views on provision of the progestogen-only pill and combined oral contraception in the pharmacy setting
Pharmacy provision of COC
When respondents were asked if their local pharmacists were currently providing COC by PGD, 86 (49.4%) said ‘no’, 18 (10.4%) ‘yes’ and 70 (40.2%) ‘unsure’.
Respondents’ views on provision of COC by pharmacists are shown in table 2. Almost half (49.1%) supported pharmacists initiating COC under PGD and the vast majority (82.0%) supported pharmacists providing COC to women continuing the method. Almost half (45.1%) opposed the reclassification of COC as a P-medicine, while over a third (39.3%) supported this. More than three-quarters of respondents (78.4%) opposed the reclassification of COC as a GSL medicine, while just over a tenth (11.7%) supported this.
Impact of pharmacy provision of POP and COC on other SRH outcomes
Respondents were asked to consider what the impact would be on six predefined SRH outcomes if POP and COC were made available in pharmacies (results shown in table 3).
Respondents’ views on the impact of pharmacy provision of the progestogen-only pill and combined oral contraception on sexual and reproductive health outcomes
Considering only those respondents who indicated ‘improve’, ‘same’ or ‘worse’, the majority (103/144, 71.5%) felt the use of bridging (temporary) contraception would improve, and 81/141 (57.4%) thought use of effective contraception would increase.
The majority of respondents (86/143, 60.1%) felt that the overall use of LARC was likely to worsen. A majority (71/130, 54.6%) felt the number of unintended pregnancies would improve (ie, decline) with the availability of POP and COC in pharmacies.
Pharmacy provision of other contraceptive methods
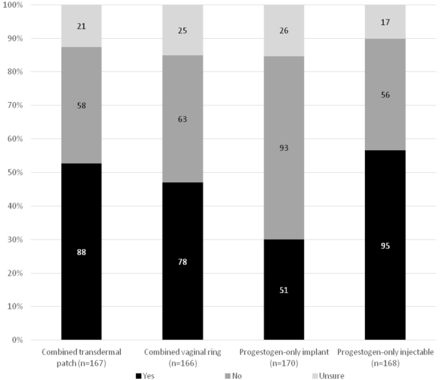
Respondents were asked whether they thought that other hormonal methods of contraception should be made available in pharmacies. The results are summarised in figure 1.
Respondents' views on pharmacy provision of other contraceptive methods.
Considering only the respondents who indicated ‘yes’ or ‘no’, most respondents supported provision of the combined transdermal patch (88/146, 60.3%), combined vaginal ring (78/141, 55.3%) and IC (95/151, 62.9%) at the pharmacy. Provision of subdermal implants was the only method that the majority of respondents (93/51, 66.0%) did not support.
Challenges and opportunities
Free-text comments from respondents provided the opportunity to raise concerns and to consolidate suggestions on how pharmacist-led provision of contraception might be implemented. Key common themes are presented below.
Access to contraception
Several respondents framed the pharmacy provision of contraception as a way to increase access and expand service delivery. This was felt to be most appropriate for women with non-complex/routine contraception requirements and could be "empowering" for individuals wanting to access contraception without "perceived barriers/judgments".
Many respondents expressed concern that the ‘contraceptive choice’ consultation could be lost, and that although access would increase, contraceptive options would be reduced. Respondents wanted pharmacists to be able to provide information about all contraceptive methods and to have robust systems in place for signposting/referring women who wanted LARC.
A strong and overarching theme was that the provision of contraception should be free. This was cited as a reason for not supporting contraception as a P-medicine or GSL. A small number of respondents reported feeling the service would not be used if patients needed to pay for it, leading to decreased access. One respondent feared that "instead of this being an additional route … it will become the ONLY route with future decommissioning of services".
Pharmacists' capacity and competency to provide contraception
Respondents expressed anxiety that pharmacists would not have sufficient time or adequate training and support to provide full, holistic sexual health consultations encompassing all methods of contraception and sexually transmitted infection (STI) screening. They expressed doubts around a pharmacist’s ability to take a thorough medical history and assess for contraindications, and whether pharmacists would have the time and skills to discuss all options and provide counselling for the woman's chosen contraceptive method. Respondents wanted pharmacists to provide information about all methods of contraception and have robust systems in place for signposting/referring women who wanted LARC or required specialist review due to medical comorbidities.
Respondents commented that some pharmacists already provided a variety of SRH services, such as EC and STI testing kits. It was suggested that those who already provided EC were in a good position to quick-start contraception after EC. In addition, if contraception provision was by PGD, then information about screening and LARC could be standardised within the PGD.
Safeguarding women
Concerns around safeguarding vulnerable women were highlighted and the need for pharmacists to have clear and robust referral pathways for these women.
Ten respondents indicated they felt that pharmacists not having access to the patient’s records, and GPs not having a record of contraception initiated by pharmacists, would be problematic. A small number were concerned that patients who had previously been advised they were unsuitable for COC could purposefully provide an incomplete health history in order to access COC from the pharmacy. It was suggested that there should be a system in place to ensure good communication between all contraception providers.
Discussion
Our study showed that the majority of respondents who were delegates at national and regional UK SRH meetings were largely supportive of pharmacist provision of hormonal contraception. Unsurprisingly, due to its safety profile and feasibility of dispensing, provision of POP was most strongly supported. This was for both initiation and continuation under PGD, and reclassification of POP to a P-medicine.
The majority of respondents also supported pharmacist-led provision of IC. A Health Services research study from Scotland also suggested that pharmacists trained to deliver repeat injections of the subcutaneous IC were enthusiastic about expanding their role to provide this but that delivery of the service could only be feasible and sustainable if a sufficiently large pool of pharmacists were trained to deliver this method.20
Although pharmacist provision of repeat COC supplies was strongly supported, respondents were more cautious about initiation of COC and reclassification to a P-medicine or GSL. This was due to safety concerns, as respondents feared pharmacists would not have adequate training and time to take a thorough medical history and to counsel women on how to use COC.
A previous evaluation of a pharmacy-delivered OC service reviewed the consultation times for pharmacists supplying OC under PGD. The mean consultation time was 19 min,12 akin to an SRH clinic appointment time and longer than the average general practice consultation. This may help alleviate HCPs fears that pharmacy consultations would be ‘rushed’, however pharmacists themselves need to be consulted to know whether or not this is feasible within their workload. One previous Scottish study surveying attitudes of community pharmacists towards provision of SRH services revealed that the majority felt that while their workload would increase, they were generally enthusiastic about expanding their role to provide more SRH services.15
Additionally, HCPs expressed concerns that pharmacists would be unable to thoroughly assess women for contraindications, is in spite of evidence showing that women are able to accurately self-screen for contraindications when a medical questionnaire is provided.21–24 In addition, respondents’ concerns that women may misinform pharmacists in order to access their preferred method of contraception may also be exaggerated. In a separate consultation with the General Pharmaceutical Council, the Faculty of Sexual & Reproductive Healthcare (FSRH) has acknowledged that although this can be an issue with any type of consultation with any HCP, women should be trusted to make decisions around their own health.25
Training for pharmacists was highlighted as a key concern, although training and assessment packages to equip pharmacists with the skills and knowledge required to issue OC under PGD are already in existence.12–14 Online learning packages, with local clinic placements for practical experience, could be used for training, with ongoing support and involvement in educational events from local services.
A collaborative relationship with local SRH services and GPs will be important if pharmacy provision is to be fully integrated into SRH services. As well as initial training and ongoing education, support mechanisms and clear referral pathways will be imperative in ensuring consistent care for patients and specialist support for pharmacists. It is therefore very promising that this study shows HCPs largely support increasing provision of contraception at pharmacies.
It was clear many respondents felt strongly that contraception should always be free. This was mirrored in free-text comments and opposition to the availability of buying contraception without a prescription. Although clearly costs could be prohibitive for some, many women in the UK are already choosing to pay for contraception by accessing it through online pharmacies. Worldwide, OCs are available for sale over the counter in a number of countries,26 and studies showing the acceptability of this scenario provide further evidence that some women find the benefit of convenience outweighs the financial cost.27 28
This study was limited as it only explored the viewpoints of HCPs. Further studies of pharmacist and patient viewpoints would be beneficial to better understand the support for pharmacy provision of contraception.
Acknowledgments
The authors would like to thank the Faculty of Sexual & Reproductive Healthcare (FSRH) and NHS Lothian for permission to conduct the research at their events. The authors would also like to thank Sally Rafie and colleagues for kindly sharing the questionnaire used in their research that helped inform the development of the questionnaire used in this study.
References
Footnotes
Contributors All authors contributed to the development of the research idea and questionnaire. KB and ZEC collected and analysed the data. All authors contributed to the preparation and review of the manuscript.
Funding The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests None declared.
Patient consent for publication Not required
Ethics approval Formal ethics approval was not sought. Permissions to conduct the research was granted by the event organisers (FSRH and NHS Lothian).
Provenance and peer review Not commissioned; externally peer reviewed.
Data availability statement Additional unpublished data from this study are unavailable as these may identify participants.