Article Text
Statistics from Altmetric.com
Key messages
UK lockdown and travel restrictions introduced in March 2020 limited access to abortion as no local services existed despite abortion being decriminalised in 2019.
An emergency regional early medical abortion (EMA) service was quickly established, is working well and patient feedback is excellent.
The future of abortion in Northern Ireland (NI) including EMA is uncertain without funding from the Department of Health NI.
Background
The 1967 Abortion Act establishing legal abortion in the rest of the UK has never applied in Northern Ireland (NI). With few exceptions, abortion has remained illegal and unavailable. Women have had to travel to Great Britain, funding treatment themselves until 2017 when the UK Department of Health and Social Care agreed to fund abortions provided by the main UK private abortion providers via a Central Booking System.
Abortion was decriminalised in NI on 22 October 2019 under the Northern Ireland (Executive Formation etc) Act 2019.1 Regulations have been introduced; however, services have yet to be commissioned.
Why was this emergency service needed?
The legal framework, The Abortion (Northern Ireland) (No. 2) Regulations 2020,2 came into effect on 31 March 2020 just after UK lockdown due to the COVID-19 pandemic, ‘stay home’ advice and the introduction of travel restrictions within the UK. Flights were cancelled, meaning accessing abortion in England involved an 8-hour ferry trip each way. Two women attempted suicide after flights were cancelled and they were unable to travel. Accessing abortion was problematic despite it being recognised as an essential service during the pandemic by the Faculty of Sexual & Reproductive Healthcare (FSRH)3 and the Royal College of Obstetricians & Gynaecologists (RCOG).
What is the law regarding EMA in NI?
Abortion is decriminalised and allowed without conditionality up to 11+6 weeks’ gestation.
For EMA (up to 9+6 weeks) mifepristone must be taken on National Health Service (NHS) premises but misoprostol can be used at home. The NI Executive failed to authorise home use of mifepristone (available elsewhere in the UK and Ireland). This would have minimised travel, reduced the risk of COVID-19 transmission and allowed local abortion access for women self-isolating or shielding.
Certification is required by one healthcare professional up to 12 weeks’ gestation.
Notification must be made to the Chief Medical Officer (CMO) NI within 14 days.
How was the service established?
The Northern Ireland Abortion and Contraception Task Group (NIACT), a multidisciplinary group, was established to provide input into the commissioning, development and implementation of an abortion service. The first meeting was held on 30 March 2020 to plan a temporary EMA service within existing sexual and reproductive health (SRH) services in response to the pandemic. Implementation of an abortion service quickly without funding was not straightforward and involved immense effort and commitment by a multidisciplinary team including medical, nursing, clerical and pharmacy staff.
Support was offered by the FSRH, RCOG and senior management of two of five Healthcare Trusts. Initial plans were halted following a message to cease and desist from the Department of Health (DoH) NI but on 9 April 2020 the CMO sent a letter permitting the Trusts to proceed.
Most staff had experience in abortion care from training elsewhere in the UK and online training resources were utilised. Clinic protocols, policies and patient information leaflets were quickly produced in line with the RCOG/National Institute for Health and Care Excellence (NICE) guideline on abortion care.4 Templates for telephone consultations and treatment were designed for the online system, Lilie.
A referral pathway was established in partnership with Informing Choices NI (ICNI), a charity that agreed to be the Central Access Point facilitating self-referral, initial assessment and safeguarding. ICNI refer daily into each Trust via email, a telephone consultation is offered within days and a time is then agreed to attend for treatment.
Experience so far – the first 3 months
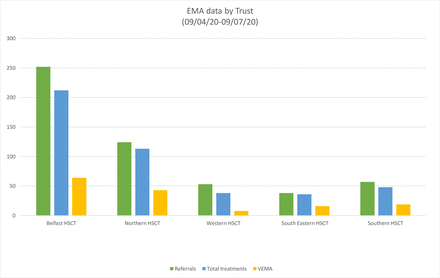
Services initially started in two Trusts. By 8 weeks there was a regional EMA service within all five Trusts operating at least two sessions per week (figure 1).
Early medical abortion data by Northern Ireland Health and Social Care Trust (HSCT). VEMA, very early medical abortion.
A total of 572 clients have contacted the ICNI Central Access Point; 524 women have had a consultation, the majority receiving EMA treatment.
One-third (150) of the women have been treated as very early medical abortion (VEMA) allowing treatment as soon as pregnancy is confirmed without ultrasound evidence of an intrauterine pregnancy.5 This a relatively new concept in the UK and has been possible due to prompt referral and short waiting times. Early treatment is safer and associated with less pain and bleeding.
Scanning is available in three clinics and elsewhere scans are available via the early pregnancy service if needed.
There have been no reported serious complications of treatment.
Contraception is offered at all clinics including immediate insertion of either a subdermal contraceptive implant or intrauterine contraception once successful treatment is confirmed. The uptake of long-acting reversible contraception (LARC) is high.
Feedback from the patient survey has been exceptionally positive. Patients are delighted to access the service locally and be offered effective contraception.
An advantage of the pandemic has been the absence of anti-choice protests outside the clinics as the facility to set up exclusion zones has not been included in the Regulations. As lockdown eases this could become a significant issue.
The Belfast Trust – in focus
The Belfast Trust has received the largest number of referrals (252; 48.0%). Of these, 212 (84%) received EMA treatment. The average waiting time was 1.58 days and the average time between consultation and treatment was 0.81 days. Average age of the women was 28.9 years. Some 126 (59.4%) women were parous and 86 (40.6%) were nulliparous. Eighty-six (40.6%) had an ultrasound, most commonly for conception while on hormonal contraception (30; 34.9%) or uncertain last menstrual period (LMP) (23; 26.7%). As per the regulations, all patients received mifepristone at the clinic and misoprostol at home. All were less than 10 weeks’ gestation. Sixty-four (30.2%) were less than 6 weeks’ gestation (VEMA). Half the women (108; 50.9%) opted for LARC either by subdermal implant (57; 26.9%) or intrauterine contraception (51; 24.0%).
The future – where do we go from here?
Demand for EMA remains high with at least 50 clients a week contacting ICNI Central Access Point. However, SRH services are being restored following suspension during the coronavirus lockdow, reducing capacity to deliver EMA. Existing contraception services are already understaffed and underfunded. The EMA staff are dedicated and conscientiously committed to providing abortion care but there has been no funding from the DoH NI. The Northern Trust withdrew their EMA service in early October, and their patients now have to travel to England. ICNI will be unable to sustain the Central Access Point without funding.
The regulations allow for provision of EMA in primary care and many general practitioners are keen to prescribe but there are issues with indemnity, prescriptions and opposition from colleagues.
Mifepristone at home is not permitted, there is no surgical option and no service for women above 9+6 weeks’ gestation hence many still need to travel to access abortion care, now including all Northern Trust patients.
Abortion remains hugely stigmatised with considerable political resistance. There is no sign of the DoH commissioning the comprehensive service that is urgently needed.
Abortion is now legal in NI and we have come a long way; unfortunately, however, safe local treatment remains inaccessible to many of those who need it.
Data availability statement
Data were provided by each Health and Social Care Trust and are available on request.
Ethics statements
Patient consent for publication
Footnotes
Twitter @sandradww46
Contributors All the authors contributed to providing and analysing data and writing this article.
Funding The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests None declared.
Patient and public involvement Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Provenance and peer review Not commissioned; externally peer reviewed.